Medical device packaging isn’t just a container. It’s part of the product.
The materials, construction, sealing, and labeling all affect whether a device performs as intended, survives the supply chain, and meets the regulatory requirements that govern its use. Get any of those wrong, and you’re not just dealing with a packaging problem.
For medical OEMs and healthcare product companies, choosing the right contract converting partner for packaging is one of the more consequential decisions in the production process. Here’s what that decision should come down to.
Why Medical Packaging Is a Different Category
Consumer packaging tolerates a range of imperfections. Medical packaging doesn’t. Dimensional accuracy, material compatibility, seal integrity, and print legibility all have to be right, every time, across every production run.
Skin-contact products carry their own specific requirements. Adhesive and barrier layers need to perform without causing irritation or degrading over the product’s shelf life.
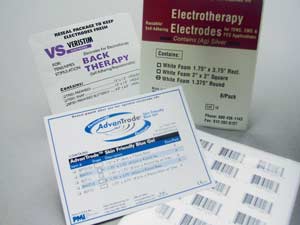
Electrotherapy electrodes, diagnostic patches, wearable monitoring sensors, and disposable clinical components each place different demands on the materials and converting processes used to package them.
Contamination control matters throughout. A finished electrode or diagnostic component that performs correctly in manufacturing can fail in the field if the packaging process introduces particulates or compromises the adhesive layer.
That’s why meticulously clean environment, foreign particulate control, and trained personnel aren’t optional in a medical converting environment. They’re baseline requirements.
What Precision Converting Looks Like in Practice
Contract converting for medical packaging involves multiple precision processes working together to create reliable, compliant products.
Key processes include:
- Rotary die cutting
High-volume production of consistent components from roll stock, commonly used for electrodes, patches, and liners. - Flatbed die cutting
Ideal for lower volumes or complex shapes that require tighter tolerances. - Multi-layer lamination
Bond materials like conductive films, foam, nonwovens, and adhesives into a single structure. Impacts peel strength, conductivity, and long-term performance. - Flexographic printing
Used for clinical labels, inserts, and backing layers. Supports durability, legibility, barcoding, and regulatory compliance.
Pepin Manufacturing brings over 30 years of experience in precision converting, including die cutting, lamination, conductive materials, hydrogel electrodes, kitting, and custom packaging.
As one medical device manufacturer shared, “Pepin understands our needs… and handles our ever-changing demands.”
That level of precision and flexibility is exactly what medical OEMs need in a contract converting partner.
Choosing the Right Materials for Medical Packaging
The materials list for medical converting is specific. Nonwoven and spunlace fabrics, medical-grade foams and films, PET and polyethylene layers, conductive carbon and silver films, skin-safe adhesive layers, and printable liners are all part of the palette.
Selecting the right combination for a given application requires experience with how those materials interact under production conditions and during extended product use.
Pepin Manufacturing works with medical-grade substrates and evaluates materials for adhesion, compatibility, and long-term wear as part of their standard process.
For products like TENS and EMS electrodes, patient monitoring pads, and custom wearable interfaces, material expertise directly affects patient comfort and device reliability.
Compliance, Documentation, and Traceability Requirements
Medical manufacturers need documentation that stands up to audits and supports full product traceability.
Key requirements include:
- Batch traceability
Tracks every product back to its source materials and production history - Raw material records
Verifies material origin, specifications, and compliance - In-process inspection logs
Document quality checks throughout production - Final approval documentation
Confirms products meet required standards before release
Pepin Manufacturing’s quality systems align with ANSI, ASTM, and CSA standards, along with RoHS and CE directives where required.
Work with a Medical Converting Partner That Meets Your Standards
Medical device converting requires a partner who understands the clinical and regulatory environment as well as the production process. Pepin Manufacturing has been that partner for companies across the healthcare industry for over 30 years.
FAQs
What is contract converting for medical devices?
Contract converting involves transforming raw materials, such as films, foams, adhesives, and conductive layers, into finished or semi-finished components through processes like die cutting, lamination, and packaging. A contract converter handles this production on behalf of the device manufacturer.
What types of medical products does Pepin Manufacturing support?
Pepin works on electrotherapy electrodes (TENS, EMS, NMES, IFC), patient monitoring pads and sensors, medical adhesive patches, disposable diagnostic components, and custom stick-to-skin wearable interfaces, among others.
What quality standards does Pepin Manufacturing follow?
Their quality systems align with ANSI, ASTM, and CSA standards, as well as RoHS and CE directives. Full batch traceability and documentation are maintained for every production run.
Can Pepin Manufacturing handle both high-volume and limited-run production?
Yes. Their just-in-time manufacturing and inventory solutions accommodate both high-volume OEM production and limited-run product lines.
Does Pepin offer private label and OEM packaging support?
Yes. They provide custom branding, branded labeling, clinical and retail pack-outs, instructional inserts, lot tracking, barcoding, and regulatory label formatting for both OEM and private-label clients.